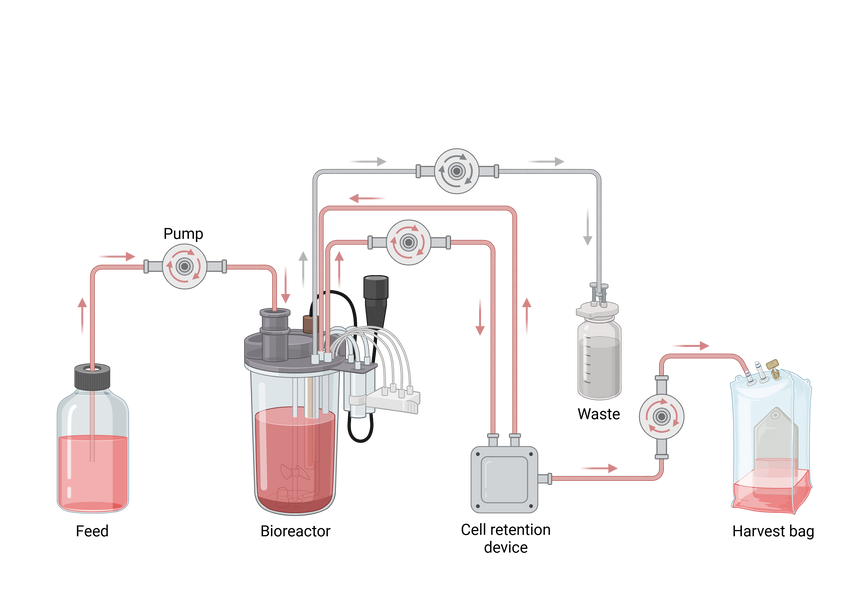
Bioreactor
7th Apr 2026
Bioreactor: Principles, Types, and Applications in Biotechnology
Introduction to Bioreactors
A bioreactor is a controlled system or vessel designed to support biologically active environments, enabling the growth of microorganisms, animal cells, or plant cells for industrial and research purposes. Bioreactors are fundamental in modern biotechnology, playing a crucial role in pharmaceuticals, food production, environmental engineering, and synthetic biology.
They provide optimal conditions such as temperature, pH, oxygen levels, and nutrient supply to maximize biological activity and product yield.
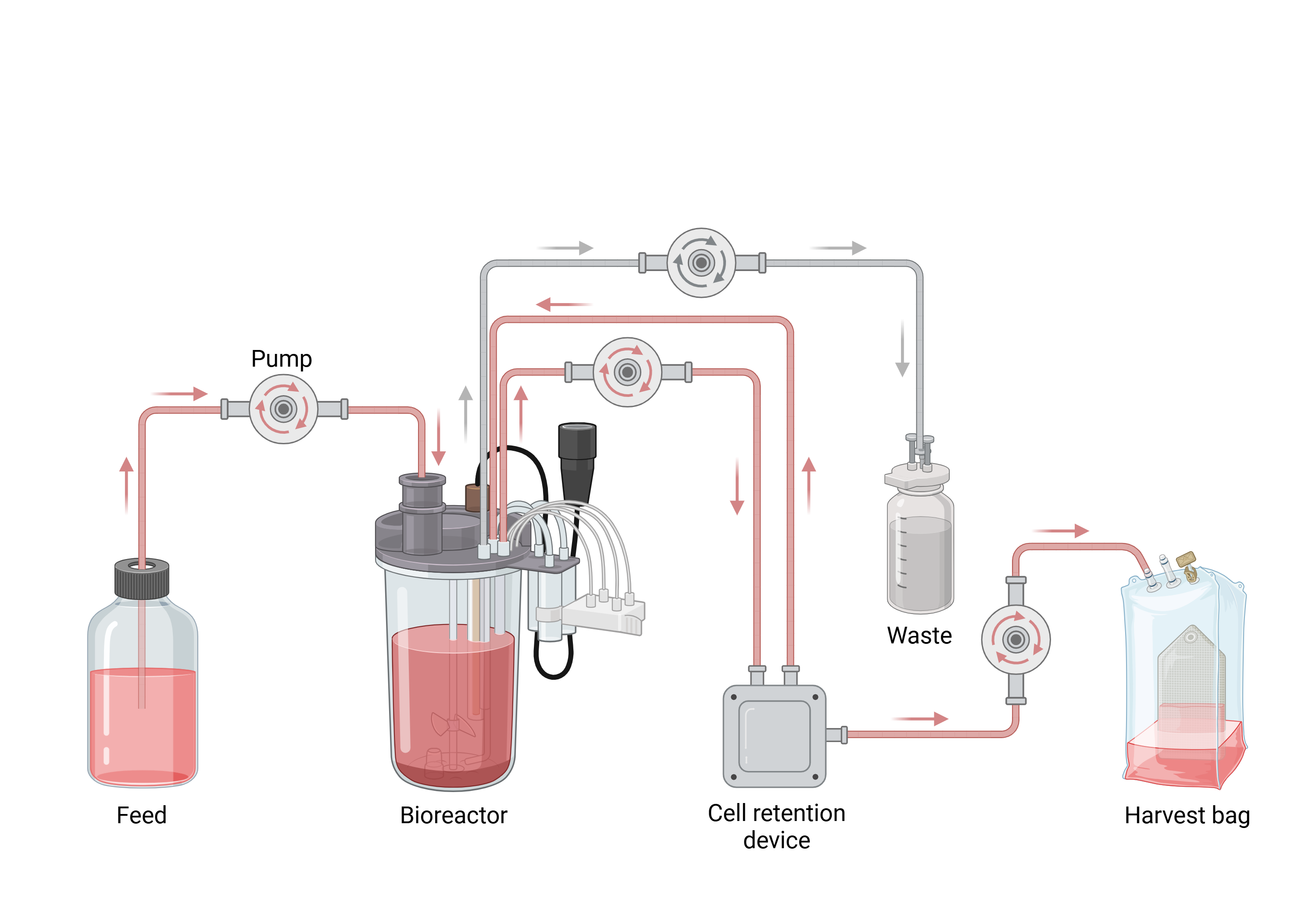
How a Bioreactor Works
Bioreactors operate based on the principle of maintaining a controlled environment that supports cellular metabolism and product formation. Key components include:
- Vessel (fermenter): The main chamber where cells are cultivated
- Agitation system: Ensures proper mixing and nutrient distribution
- Aeration system: Supplies oxygen for aerobic processes
- Sensors and control units: Monitor pH, temperature, dissolved oxygen, and pressure
- Sterilization system: Prevents contamination
The efficiency of a bioreactor depends heavily on maintaining these parameters within optimal ranges.
Types of Bioreactors
Bioreactors are classified based on their design and mode of operation:
1. Batch Bioreactor
- Operates as a closed system
- All nutrients are added at the beginning
- Widely used in laboratory-scale experiments
2. Fed-Batch Bioreactor
- Nutrients are added continuously or intermittently
- Prevents substrate inhibition
- Common in industrial enzyme and antibiotic production
3. Continuous Bioreactor
- Fresh medium is continuously added while culture is removed
- Maintains steady-state conditions
- Used for large-scale industrial processes
4. Stirred Tank Bioreactor (STR)
- Most commonly used type
- Equipped with impellers for mixing
- Suitable for microbial and mammalian cell cultures
5. Airlift Bioreactor
- Uses air bubbles for mixing instead of mechanical agitation
- Energy-efficient and gentle on shear-sensitive cells
6. Packed Bed Bioreactor
- Contains immobilized cells on solid supports
- Used in wastewater treatment and biocatalysis
Key Parameters in Bioreactor Operation
To ensure optimal performance, several critical parameters must be tightly controlled:
- Temperature: Influences enzyme activity and cell growth
- pH: Affects metabolic processes and product stability
- Dissolved Oxygen (DO): Essential for aerobic organisms
- Agitation speed: Impacts mixing and oxygen transfer
- Nutrient concentration: Determines growth rate and productivity
Precise monitoring and automation systems are often employed to maintain these parameters.
Applications of Bioreactors
Bioreactors have a wide range of applications across industries:
1. Pharmaceutical Industry
- Production of vaccines, monoclonal antibodies, and recombinant proteins
- Example: insulin production using genetically engineered bacteria
2. Food and Beverage Industry
- Fermentation processes
- Enzyme production for food processing
3. Environmental Biotechnology
- Wastewater treatment using microbial consortia
- Bioremediation of polluted environments
4. Biofuel Production
- Cultivation of algae and microorganisms for bioethanol and biodiesel
5. Tissue Engineering
- Growth of artificial tissues and organs under controlled conditions
Advantages and Limitations
Advantages
- High productivity and scalability
- Controlled and reproducible conditions
- Efficient use of biological systems
Limitations
- High initial investment cost
- Risk of contamination
- Complex operation and maintenance
Future Trends in Bioreactor Technology
Advances in biotechnology are driving innovation in bioreactor design:
- Single-use bioreactors for reduced contamination risk
- Smart bioreactors with AI-driven monitoring systems
- Integration with Big Data and cloud computing for process optimization
- Development of microbioreactors for high-throughput screening
These innovations are enhancing efficiency, flexibility, and scalability in bioprocessing.
Conclusion
Bioreactors are indispensable tools in modern science and industry, enabling the large-scale production of valuable biological products. With continuous technological advancements, bioreactors are becoming more efficient, automated, and adaptable to diverse applications from pharmaceuticals to environmental sustainability.