EPA Method 9013A
The toxicity and mobility of cyanide in soil is governed by its chemical form. Simple cyanide, or the cyanide ion (CN- ), can be weakly adsorbed onto soil particles at pH>9.2. Weak metal-cyanide complexes ([M(CN4)]-2) and strong metal – cyanide complexes ([M(CN)6]-3 or [M(CN)6]-4) have an affinity for metal oxides and organic matter that decreases with increasing pH, however, other salts in solution tend to inhibit adsorption1.
Simple cyanide, weak metal-cyanide complexes, and strong-metal cyanide complexes are readily soluble in water. Metal – Metal cyanide complexes, such as Prussian Blue (Fe4[Fe(CN)6]3), are insoluble in water and are the most common forms of cyanide found in sediment and soil2. Metal-metal cyanide complexes are insoluble in acid solution, and solubility increases with pH.
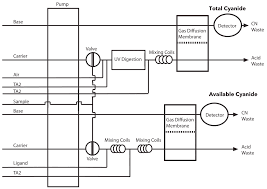
A common practice for the extraction of cyanide in soil and sediment is acid distillation3. This approach is valid for all cyanide forms except the metal-metal cyanide complexes that are most likely to be present.
The quality control practice of spiking a soil sample with simple cyanides, or strong metal-cyanide complexes to validate acid distillation is misleading because these cyanide complexes are readily soluble. Acid distillation of cyanide in soil results in low and irreproducible recoveries for the metal-metal cyanide complexes most likely to be present. Acid distillation of cyanide in soil is ineffective and does not accurately measure “total” cyanide nor does it estimate cyanide toxicity.
There are no products listed under this category.