Host Cell DNA: A Comprehensive Guide to Residual DNA in Biopharmaceuticals
23rd Mar 2026
Host Cell DNA: A Comprehensive Guide to Residual DNA in Biopharmaceuticals
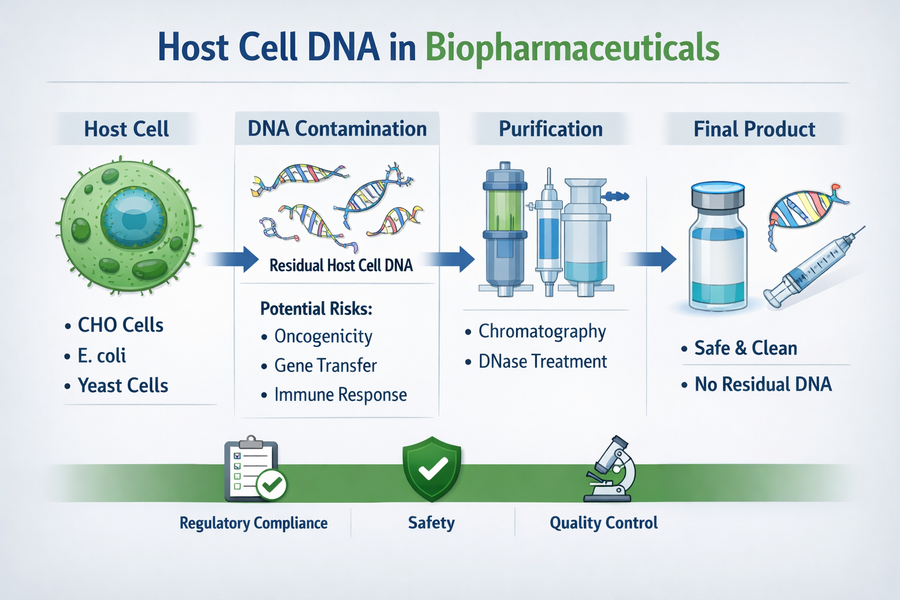
Host cell DNA is a critical quality attribute in the production of biopharmaceuticals, vaccines, and recombinant proteins. During the manufacturing process, therapeutic proteins are expressed in living systems such as bacteria, yeast, or mammalian cells. As a result, residual host cell dna can remain in the final product and must be carefully monitored and controlled to ensure product safety and regulatory compliance.
What is Host Cell DNA?
Host cell dna refers to the genetic material originating from the cells used to produce recombinant biologics. These host cells may include:
- Bacterial systems (e.g., Escherichia coli)
- Yeast cells (e.g., Saccharomyces cerevisiae)
- Mammalian cells (e.g., Chinese Hamster Ovary (CHO) cells)
During upstream processing and cell lysis, DNA from these cells is released into the production environment. Despite downstream purification steps, trace amounts of host cell dna may persist in the final therapeutic product.
Why is Host Cell DNA Important?
Residual host cell dna is considered a process-related impurity and poses potential risks:
1. Safety Concerns
- Possible oncogenicity due to DNA fragments
- Risk of horizontal gene transfer
- Potential immunogenic responses
2. Regulatory Requirements
Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency impose strict limits on host cell dna levels in biologics.
Typical guidelines recommend:
- Less than 10 ng of DNA per dose
- DNA fragment size reduced to <200 base pairs
Sources of Host Cell DNA Contamination
Residual host cell dna can originate from several stages of biopharmaceutical production:
- Cell lysis during protein extraction
- Incomplete DNA removal during purification
- Inefficient enzymatic digestion (e.g., DNase treatment)
- Carryover from upstream processing
Understanding these sources is essential for designing effective removal strategies.
Methods for Detection and Quantification
Accurate detection of host cell dna is essential for quality control. Common analytical methods include:
1. Quantitative PCR (qPCR)
- Highly sensitive and specific
- Detects low levels of residual DNA
- Requires standard curves for quantification
2. Digital PCR (dPCR)
- Provides absolute quantification
- Higher precision for low-abundance DNA
- Ideal for trace-level detection
3. Fluorescent DNA Binding Assays
- Use dyes such as PicoGreen
- Rapid but less specific than PCR-based methods
4. Next-Generation Sequencing (NGS)
- Advanced method for DNA characterization
- Can identify sequence origin and fragment size
Strategies for Removal of Host Cell DNA
Efficient removal of host cell dna is a key objective in downstream processing:
1. Enzymatic Digestion
- DNases are used to fragment DNA into smaller pieces
- Enhances removal during filtration
2. Chromatography Techniques
- Ion-exchange chromatography removes negatively charged DNA
- Affinity chromatography improves purification efficiency
3. Filtration and Ultrafiltration
- Size-based separation removes DNA fragments
- Often combined with other purification steps
4. Precipitation Methods
- Chemical precipitation of DNA using salts or alcohols
A combination of these strategies ensures minimal residual contamination.
Regulatory Guidelines for Host Cell DNA
Global regulatory agencies emphasize strict control of host cell dna:
- Limits on total DNA content
- Control of DNA fragment size
- Validation of removal processes
- Routine testing during manufacturing
Compliance with guidelines from organizations such as the World Health Organization is essential for product approval.
Applications and Relevance in Biopharmaceuticals
Monitoring host cell dna is crucial in:
- Monoclonal antibody production
- Recombinant protein therapeutics
- Vaccine manufacturing
- Gene therapy products
In these applications, ensuring minimal DNA contamination is essential for patient safety and product efficacy.
Challenges in Host Cell DNA Analysis
Despite technological advances, several challenges remain:
- Detecting ultra-low levels of DNA
- Differentiating host DNA from product-related nucleic acids
- Standardization of analytical methods
- Ensuring reproducibility across laboratories
Future Perspectives
The future of host cell dna analysis is evolving with:
- Integration of digital PCR and NGS technologies
- Automation of quality control workflows
- Development of more sensitive detection assays
- Improved purification technologies
These advancements will enhance safety and efficiency in biopharmaceutical manufacturing.
Conclusion
Host cell dna is a critical impurity that must be carefully monitored and controlled in biologics production. Through advanced detection methods and robust purification strategies, manufacturers can ensure compliance with regulatory standards and deliver safe, high-quality therapeutic products. As analytical technologies continue to evolve, the management of host cell dna will become even more precise and reliable.