Principle of HPLC
23rd Mar 2026
Principle of HPLC: A Comprehensive Scientific Overview
High-Performance Liquid Chromatography (HPLC) is a cornerstone technique in analytical chemistry, biochemistry, and pharmaceutical sciences. Its precision, sensitivity, and reproducibility make it indispensable for the analysis of complex mixtures. Understanding the principle of HPLC is critical for researchers, students, and laboratory professionals who rely on chromatographic techniques for qualitative and quantitative analysis.
What is HPLC?
HPLC is a chromatographic technique that separates components in a mixture based on their interaction with a stationary phase and a mobile phase. Unlike traditional column chromatography, HPLC uses high-pressure pumps to force the solvent through densely packed columns, increasing separation efficiency and resolution.
HPLC can analyze a wide range of compounds, including:
- Small organic molecules
- Peptides and proteins
- Nucleic acids
- Pharmaceuticals and metabolites
This versatility makes it an essential tool in research, quality control, and clinical diagnostics.
The Principle of HPLC
The principle of HPLC is founded on the differential interaction between the analytes and the two phases in the system: the mobile phase and the stationary phase.
1. Mobile Phase
The mobile phase is a liquid solvent that carries the sample through the column. Its composition can be tailored (single solvent or mixture) depending on the polarity and chemical properties of the analytes.
- Aqueous solutions: Commonly used in reverse-phase HPLC
- Organic solvents: Methanol, acetonitrile, and buffer mixtures
- The choice of mobile phase directly affects retention time, resolution, and peak shape
2. Stationary Phase
The stationary phase is a solid material, typically silica or polymer beads, packed inside the HPLC column. The chemical properties of the stationary phase determine how strongly compounds are retained.
- Polar stationary phase → used in normal-phase HPLC
- Non-polar stationary phase → used in reverse-phase HPLC
- Special stationary phases → ion-exchange, size-exclusion, and chiral HPLC
3. Separation Mechanism
HPLC separation occurs because each compound in the mixture interacts differently with the stationary phase.
- Strong interaction: Molecules spend more time in the column → longer retention time
- Weak interaction: Molecules move faster → shorter retention time
The result is a series of peaks on a chromatogram, where each peak represents a separated compound.
Modes of HPLC
HPLC can operate in several modes depending on the type of separation required:
- Normal-Phase HPLC:
- Polar stationary phase
- Non-polar mobile phase
- Best for polar compounds
- Reverse-Phase HPLC (RP-HPLC):
- Non-polar stationary phase
- Polar mobile phase
- Most common mode, ideal for pharmaceuticals and biomolecules
- Ion-Exchange HPLC:
- Separates molecules based on charge
- Frequently used for proteins, peptides, and nucleotides
- Size-Exclusion HPLC (SEC):
- Separates based on molecular size
- Widely used for proteins, polymers, and nanoparticles
- Chiral HPLC:
- Separates enantiomers using chiral stationary phases
- Important in drug development for stereoisomer analysis
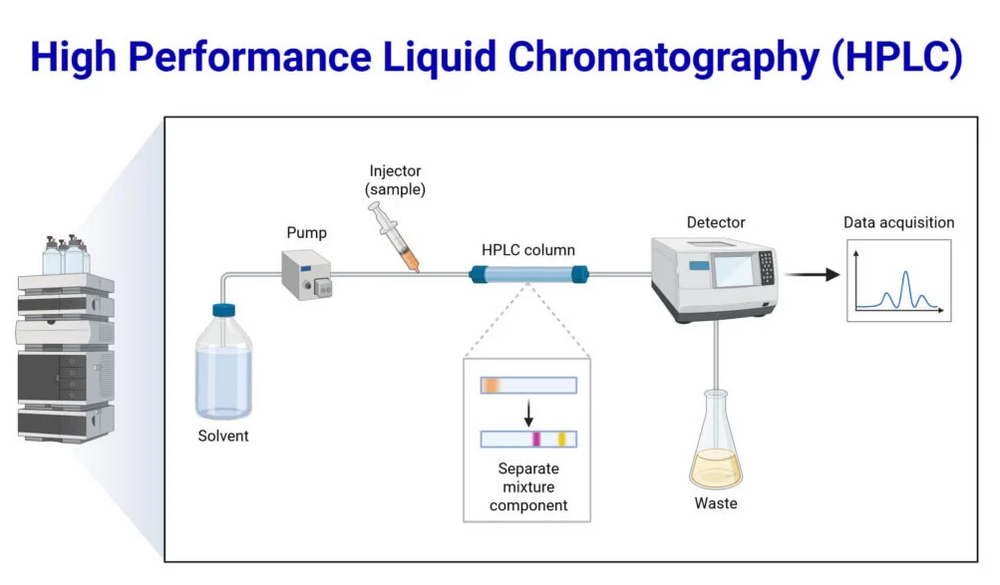
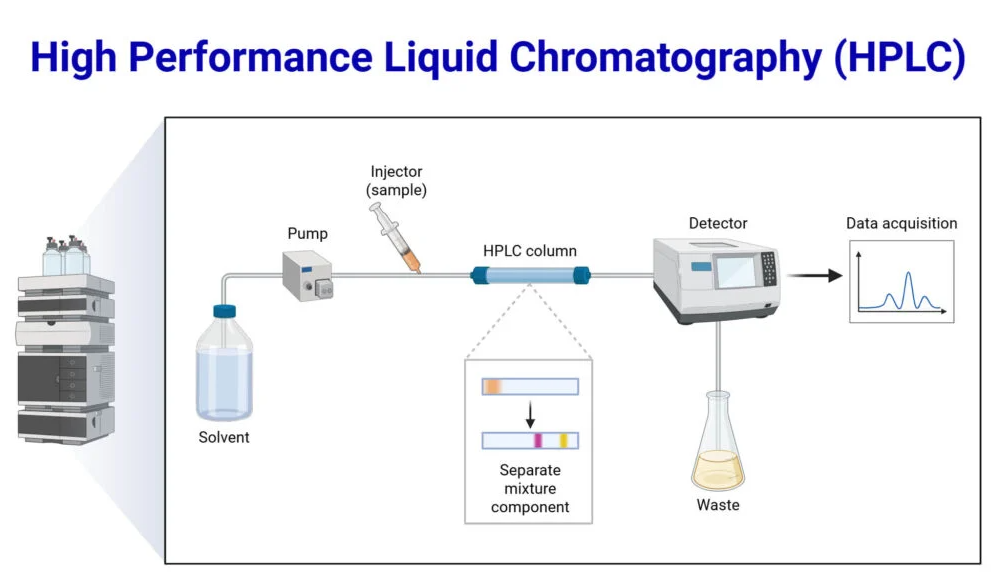
Components of an HPLC System
A modern HPLC system consists of several critical components:
- Pump: Delivers the mobile phase at high pressure, ensuring uniform flow through the column.
- Injector: Introduces a precise volume of sample into the mobile phase.
- Column: Contains the stationary phase where separation occurs.
- Detector: Measures the eluted compounds, commonly using UV-Vis, fluorescence, or mass spectrometry.
- Data System: Converts detector signals into chromatograms for quantitative analysis.
Each component must be carefully calibrated to maintain high resolution, reproducibility, and accuracy.
Applications of HPLC
The versatility of HPLC allows it to be applied in multiple fields:
- Pharmaceutical Industry: Drug purity testing, quantification, and impurity profiling
- Food and Beverage Analysis: Detection of vitamins, additives, preservatives, and contaminants
- Environmental Monitoring: Measurement of pollutants, pesticides, and heavy metals
- Clinical Diagnostics: Hormone quantification, biomarker analysis, therapeutic drug monitoring
- Biotechnology: Protein and peptide characterization, nucleic acid analysis
HPLC is particularly valuable in quality control where high precision and reproducibility are required.
Advantages of HPLC
- High resolution and accuracy
- Capable of analyzing thermally unstable or high molecular weight compounds
- Compatible with a wide range of detectors
- Rapid and reproducible separations
- Scalable from analytical to preparative levels
Limitations
While highly versatile, HPLC also has some limitations:
- Expensive equipment and maintenance
- Requires trained personnel
- Sample preparation may be time-consuming
- Not ideal for volatile compounds (requires GC for those)
Visual Representation
A simple schematic of HPLC demonstrates the principle of HPLC:
- Mobile phase enters the column under high pressure
- Sample compounds interact with the stationary phase differently
- Compounds elute at different retention times
- Detector generates a chromatogram with distinct peaks
Conclusion
Understanding the principle of HPLC is essential for accurate analysis in research, industry, and clinical laboratories. By mastering the interaction between the stationary and mobile phases, scientists can separate, identify, and quantify complex mixtures with high precision. The continuous development of HPLC methods and stationary phases ensures that it remains a cornerstone technique in analytical chemistry.